
Therefore the radius of atoms increases as you go down a. And I think that your question initially meant to compare the size of #Al^(3+)# NOT #Al^(3-)# (largely unknown).Īnd now to the anions, and cations.and for a given PERIOD, a CATION should be smaller than an anion, inasmuch as we remove an electron, and the remaining electrons should be (and are) held more tightly at closer radii due to the absence of electronic shielding. Complete answer: As we know, that an atom gets larger as its the number of shells increases. When an electron is added, a new proton is also added to the nucleus, which gives the nucleus a stronger positive charge and a greater nuclear attraction. Within a period of elements, each new electron is added to the same shell. For selenium, potassium, and bromine, we gots #n=4#.potassium should have the biggest atomic radius, followed by selenium, then bromine.i.e. The first atomic radius periodic trend is that atomic size decreases as you move left to right across a period. Than 4th row potassium, selenium, and bromine.because its valence shell is #n=3# rather than #n=4#. A 59) Why is the first ionization energy of sulfur smaller than the first ionization energy of phosphorus Answer: When sulfur loses one electron, it take on a particularly stable, half-filled p subshell. They generally decrease across each period These trends can be explained by. And this is basic and assumed knowledge for 1st year.Īnd so we repair to the Table.and we compare, #Al#, #Se#, #K#, and #Br# ATOMS.Īluminum is a third-row element.and thus it SHOULD be smaller Atomic radius Atomic radii show predictable patterns across the periodic table. 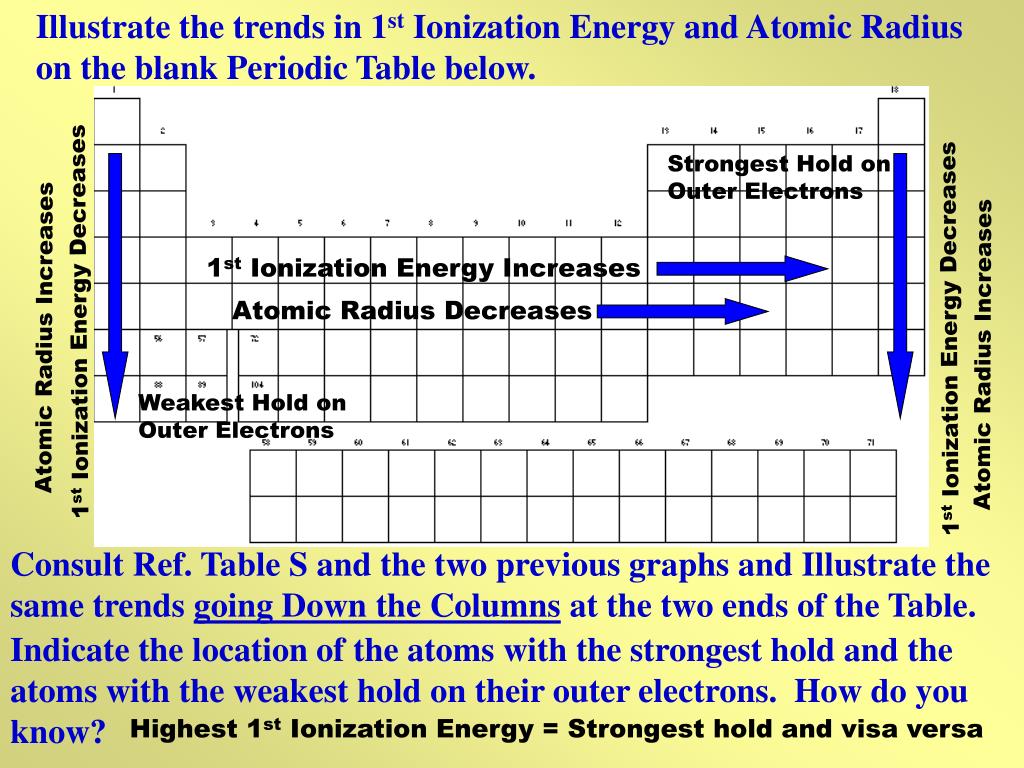
not the exoplanet has an atmosphere, but also what atoms and molecules are in it. Pages 8 This preview shows page 3 - 5 out of 8 pages. To perform its intended science meant dropping to less than 7 kelvins. School University of Alabama, Birmingham Course Title CH 115 Uploaded By JusticeSandpiper366. Place the following atoms in order of decreasing. And of course with respect to a row, a Period of the Periodic Table as we face the Table, the size atoms (defined by their valence electronic radius) should DECREASE, and that with respect to a column, a Group of the Periodic Table, atomic radii should INCREASE down the Group. Place the following atoms in order of decreasing atomic radii the largest one. 
Reasonably, we address the radii of ions and atoms, by the radius of their VALENCE electron. ANIONS should be LARGER than their parent atoms? Find an answer to your question What is the order of decreasing atomic radius 1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
